Last Updated on 1 year by Francis
Electricity is at the heart of modern technology. It powers our lights and appliances, and it even helps us to communicate with each other. But how does electricity actually work? One of the key concepts behind electricity is the idea of positive and negative charges. But which is stronger, a positive or negative charge? In this article, we’ll explore the differences between positive and negative charges, and find out which one is stronger.
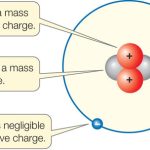
A positive charge is stronger than a negative charge. This is due to the fact that protons, which are positively charged, are heavier than electrons, which are negatively charged. This means that a positively charged particle will have a greater force of attraction than a negatively charged particle. The difference in the strength of the charges is measured in terms of electrostatic potential energy. The higher the electrostatic potential energy, the greater the difference in the forces of attraction between two particles.
Contents
What is a Positive or Negative Charge?
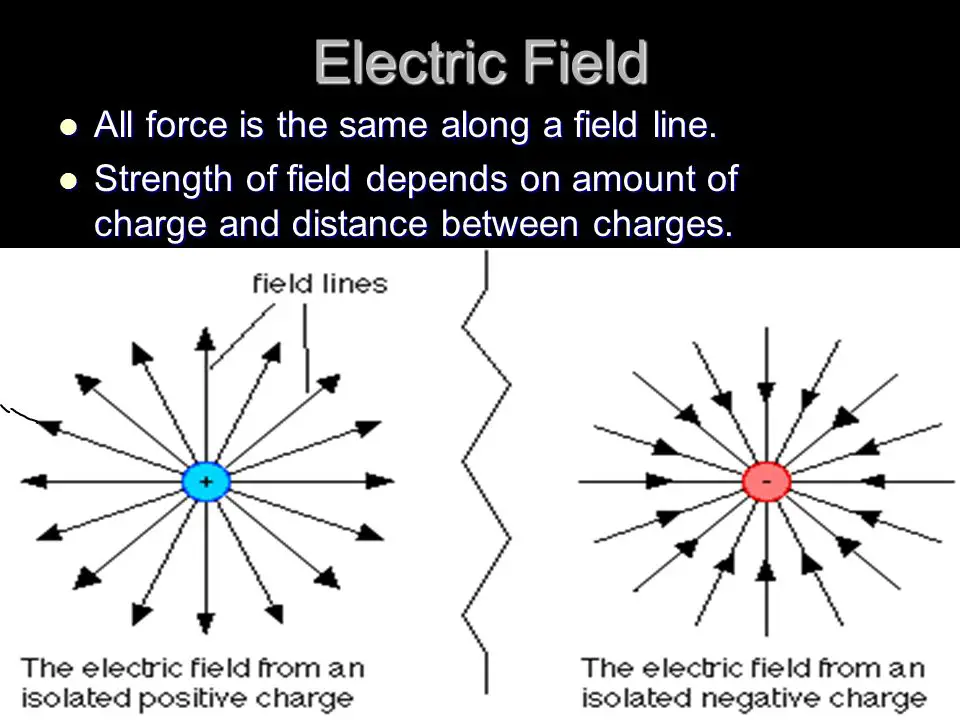
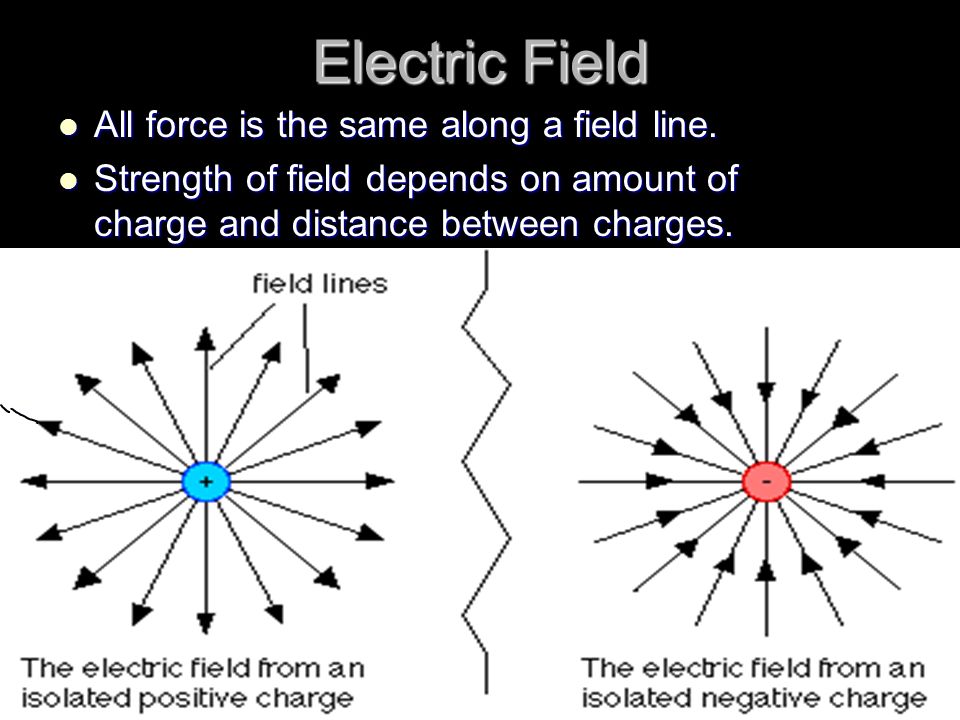
A charge is a property of matter that causes it to experience a force when placed in an electromagnetic field. Charges come in two varieties: positive and negative. Positive charges are called protons and are found in the nucleus of atoms, while negative charges are called electrons and are found in the outer shell of atoms.
The charge of an atom is determined by the number of protons and electrons it has. If an atom has more protons than electrons, it has a positive charge, and if it has more electrons than protons, it has a negative charge.
The strength of a charge is determined by the number of protons and electrons it has. The more protons and electrons an atom has, the stronger the charge will be.
What is the Difference Between a Positive and Negative Charge?
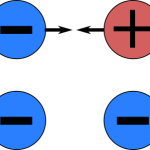
The main difference between a positive and negative charge is their direction. A positive charge will repel other positive charges, while a negative charge will attract other negative charges. This is due to the fact that like charges repel, while unlike charges attract.
Another difference between a positive and negative charge is the amount of energy they possess. Positive charges have more energy than negative charges, meaning that they are more likely to move and interact with other objects.
Lastly, a positive charge is more likely to stay in one place, while a negative charge is more likely to move around. This is because positive charges are attracted to negative charges, so they will move towards them.
Which is Stronger a Positive or Negative Charge?
When it comes to the strength of a charge, the answer is not clear-cut. In general, a positive charge is more powerful than a negative charge, but there are exceptions.
The strength of a charge depends on the number of protons and electrons it has. A charge with more protons and electrons will be stronger than one with fewer. This means that a charge with a larger number of protons and electrons will be stronger than one with a smaller number.
The Effect of Distance on Charge Strength
The distance between two charges also affects their strength. The closer two charges are, the stronger their force will be. This is because the electric field of a charge decreases with distance, so when two charges are closer together, they will experience a stronger force.
On the other hand, distance can also weaken the strength of a charge. This is because if two charges are too far apart, the electric field between them will be too weak to cause a significant force.
The Effect of Charge Type on Charge Strength
The type of charge also affects the strength of a charge. A charge with a larger number of protons and electrons will be stronger than one with a smaller number. This means that a positive charge will be stronger than a negative charge if it has more protons and electrons.
However, this does not always hold true. Depending on the distance between the charges and the type of material they are interacting with, a negative charge can be stronger than a positive one.
Conclusion
In general, a positive charge is stronger than a negative charge, but this is not always the case. The strength of a charge depends on the number of protons and electrons it has, the distance between the charges, and the type of material they are interacting with.
Top 6 Frequently Asked Questions
What is a Positive Charge?
A positive charge is an electrical charge that has more protons than electrons. It is the opposite of a negative charge, which has more electrons than protons. Positive charges typically move away from negative charges and vice versa. Positive charges can also attract negative charges, creating an electrostatic force. This force can be used to power electrical equipment such as motors, generators and light bulbs.
What is a Negative Charge?
A negative charge is an electrical charge that has more electrons than protons. It is the opposite of a positive charge, which has more protons than electrons. Negative charges typically move towards positive charges and vice versa. Negative charges can also repel other negative charges, creating an electrostatic force. This force can be used to power electrical equipment such as motors, generators and light bulbs.
Which is Stronger a Positive or Negative Charge?
In general, a negative charge is stronger than a positive charge. This is because the force of attraction between two negative charges is greater than the force of attraction between two positive charges. The strength of the force is determined by the amount of charge that each particle has. The more charge that each particle has, the stronger the force.
What is the Difference Between Positive and Negative Charges?
The main difference between positive and negative charges is the direction in which they move. Positive charges move away from negative charges and vice versa, while negative charges move towards positive charges and vice versa. Additionally, positive charges attract negative charges while negative charges repel other negative charges.
What is the Effect of Positive and Negative Charges?
Positive and negative charges have different effects on objects. Positive charges can attract negative charges and create an electrostatic force, while negative charges can repel other negative charges. This force can be used to power electrical equipment such as motors, generators and light bulbs.
How is a Positive or Negative Charge Created?
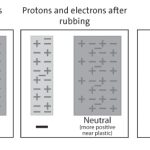
A positive or negative charge is created when an atom gains or loses electrons. When an atom gains electrons it creates a negative charge, and when an atom loses electrons it creates a positive charge. This process is known as ionization and is the result of an imbalance in the number of protons and electrons in an atom.
Positive and Negative Charge
In conclusion, the debate of whether a positive or negative charge is stronger is ongoing. Though both charges have their own unique properties, they are both capable of providing significant amounts of energy. Ultimately, it is up to the individual to decide which one is best suited for their needs. By understanding the differences between a positive and a negative charge, it is possible to make an educated decision on which one is best for the job.